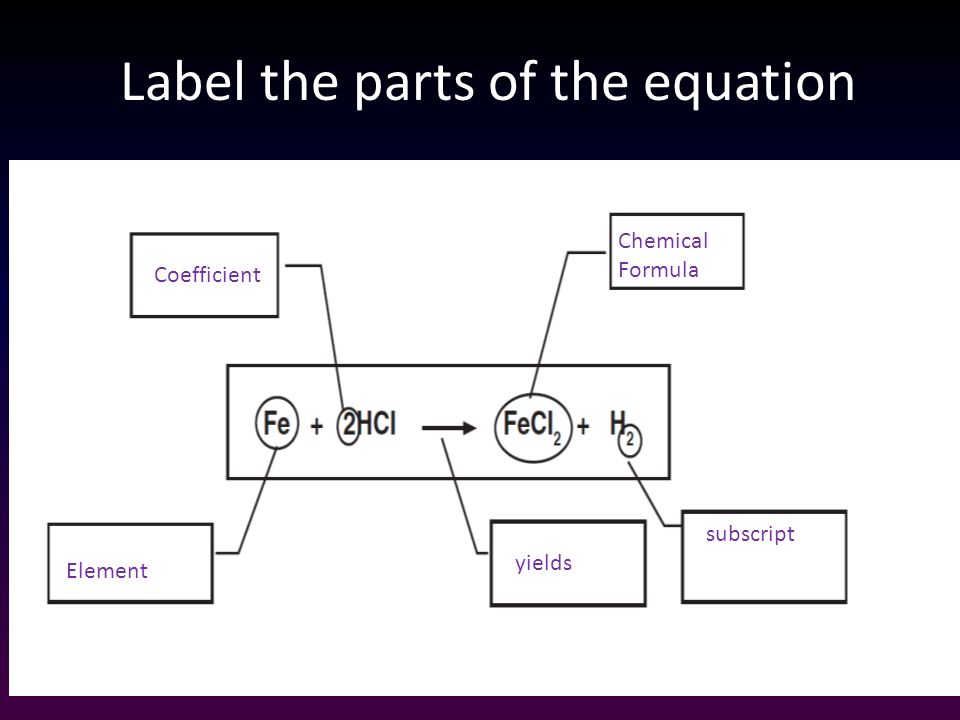
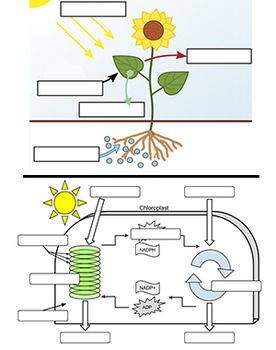
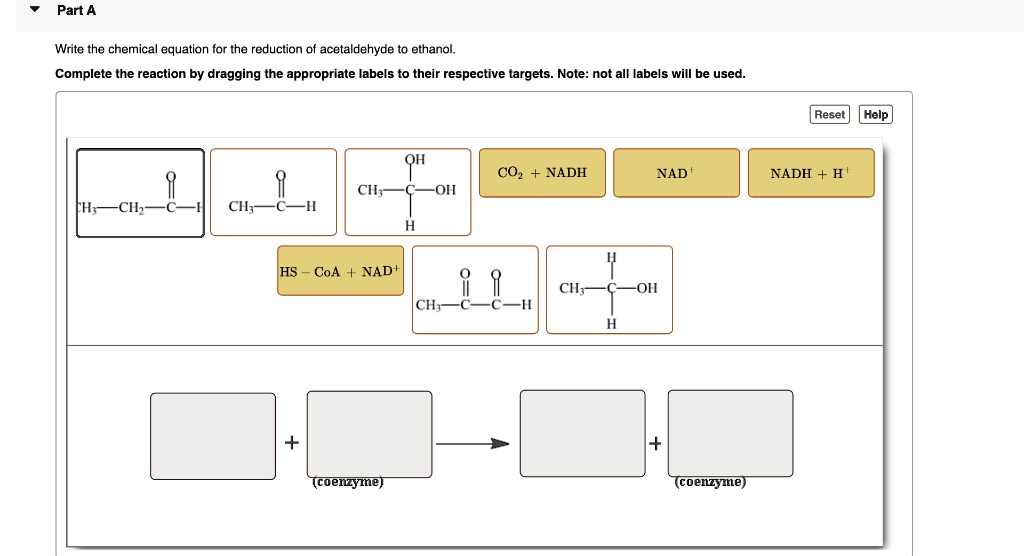
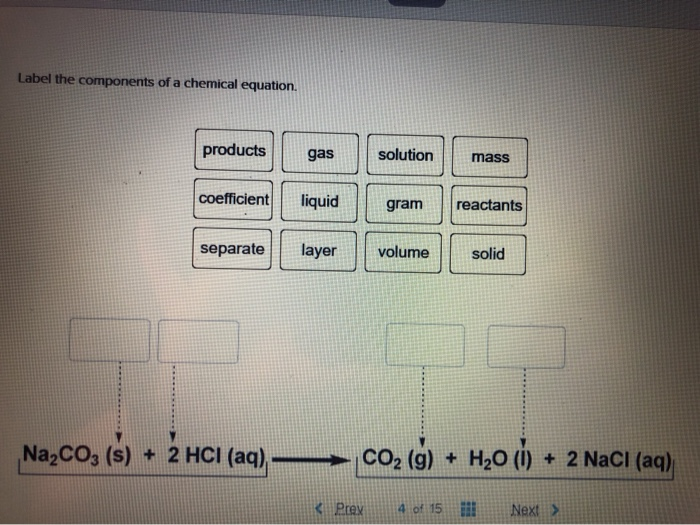
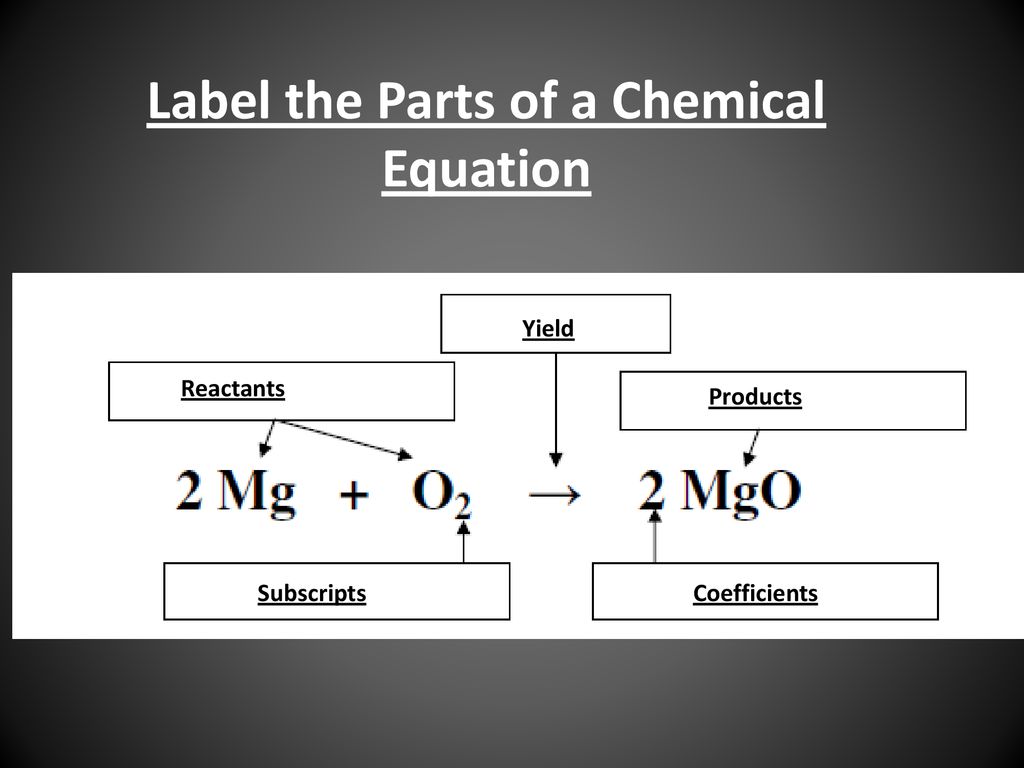
43 labels of a chemical equation
3.1: Chemical Equations - Chemistry LibreTexts When a chemist encounters a new reaction, it does not usually come with a label that shows the balanced chemical equation. Instead, the chemist must identify the reactants and products and then write them in the form of a chemical equation that may or may not be balanced as first written. Chemical equation - Wikipedia To indicate physical state of a chemical, a symbol in parentheses may be appended to its formula: (s) for a solid, (l) for a liquid, (g) for a gas, and (aq) for an aqueous solution. This is especially done when one wishes to emphasize the states or changes thereof.
Chemical Equations - Definition, Representation, Types In an equation, the products are always written on the right side, with a plus symbol (+) between them. Between the reactants and the products, an arrow sign (→) pointing to the right is placed. This arrow indicates that the substances on the left-hand side of the equation are combined to produce the substances on the right-hand side.
Labels of a chemical equation
Label the components of a chemical_ equation: - YouTube Label the components of a chemical_ equation:Watch the full video at: ... How do you label a chemical equations? - Answers Chemical equations represent chemical composition of reactants and products and also how does the reaction occur. Given chemical equations be able to balance them? When one is given the chemical ... 4.E: Chemical Reactions and Equations (Exercises) One element replaces another element in a compound. Zn + Fe (NO 3) 2 → Zn (NO 3) 2 + Fe 3F 2 + 2FeI 3 → 3I 2 + 2FeF 3 Sn + H 2 SO 4 → SnSO 4 + H 2 2Al + 3NiBr 2 → 2AlBr 3 + 3Ni No reaction occurs. Fe (NO 3) 3 + Al → Al (NO 3) 3 + Fe 2NaI + Cl 2 → 2NaCl + I 2 No reaction occurs. Zn (NO 3) 2 + 2NaOH → Zn (OH) 2 + 2NaNO 3 2HCl + Na 2 S → 2NaCl + H 2 S
Labels of a chemical equation. Label each reactant and product in the given chemical reaction. Carbon dioxide, a product of metabolism, reacts with water to release protons. Complete the chemical equation below. Identify the type of reaction shown in the following chemical equation. NH4HCO3(s) arrow NH3(g) + CO2(g) + H2O(g) Write and name the two products from a combustion reaction. Examples of Balanced Chemical Equations - ThoughtCo 6 carbon dioxide + 6 water yields 1 glucose + 6 oxygen 2 AgI + Na 2 S → Ag 2 S + 2 NaI 2 silver iodide + 1 sodium sulfide yields 1 silver sulfide + 2 sodium iodide Ba 3 N 2 + 6 H 2 O → 3 Ba (OH) 2 + 2 NH 3 3 CaCl 2 + 2 Na 3 PO 4 → Ca 3 (PO 4) 2 + 6 NaCl 4 FeS + 7 O 2 → 2 Fe 2 O 3 + 4 SO 2 PCl 5 + 4 H 2 O → H 3 PO 4 + 5 HCl 3.10: Writing and Balancing Chemical Equations When a chemist encounters a new reaction, it does not usually come with a label that shows the balanced chemical equation. Instead, the chemist must identify the reactants and products and then write them in the form of a chemical equation that may or may not be balanced as first written. How do you Write a Chemical Equation? - A Plus Topper Chemical equations give us the following qualitative information. (a) Reactants and products of a chemical reaction. (b) Physical states of the reactants and products. Take the following equation as an example. 2C(s) + O 2 (g) 2CO(g) From the equation, we know that the reactants are solid carbon and oxygen gas. The product of the reaction is ...
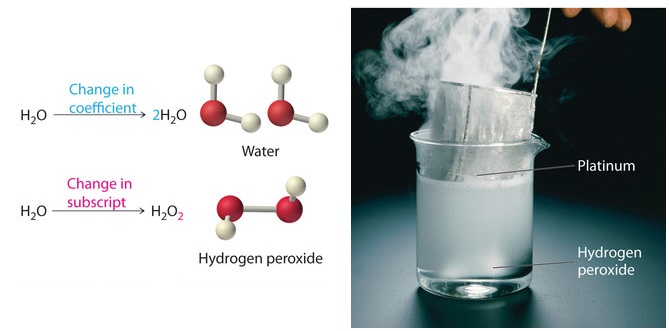
What is a Chemical Equation? - Definition & Examples We read the equation as ''A plus B yields AB'' (or ''A plus B forms AB'' or ''A plus B goes to AB''). Note that A and B are reactants, and AB is the product (a newly formed substance in a... Label the chemical Equation - Liveworksheets Label the chemical Equation worksheet Live worksheets > English Label the chemical Equation Label the parts of the chemical Equation ID: 1570824 Language: English School subject: Science Grade/level: 5 Age: 7-9 Main content: Chemical Equation Other contents: NA Add to my workbooks (0) Download file pdf Embed in my website or blog Chemical Formula Labels Teaching Resources | Teachers Pay Teachers In this worksheet, students will identify the parts of a chemical formula (coefficients, subscripts) and label the parts of a chemical equation (reactants, products, yields). On the back of the worksheet, students will count the atoms on both sides of the chemical equation to determine whether an equation is following the law of conservation of ... The Chemical Equation - Introductory Chemistry - 1st Canadian Edition Many chemical equations also include phase labels for the substances: (s) for solid, (ℓ) for liquid, (g) for gas, and (aq) for aqueous (i.e., dissolved in water). Special conditions, such as temperature, may also be listed above the arrow. For example: 2NaHCO 3 (s) → 200°C Na 2 CO 3 (s) + CO 2 (g) + H 2 O (ℓ) Key Takeaways
What are the Parts of a Chemical Equation? | Life Persona The steps followed to write a chemical equation are: - Reagents and reaction products are identified and noted. - The formula or symbols of the reagents are written on the left side with a '+' sign between them. - The formula (s) of the products are written on the right side with a '+' sign between them. Chemical Equation | Reactants And Products In Chemical Reactions - BYJUS Chemical Equation | Reactants And Products In Chemical Reactions Understand the concept of balancing chemical Equation | Law of conservation of mass, reactants and Products | Why is it important to balance a chemical equation,balanced chemical equation for magnesium oxide. Login Study Materials NCERT Solutions NCERT Solutions For Class 12 Product Labels: How to Identify Chemicals and Other Ingredients - WebMD Manufacturers can also get exemptions from labeling law for trade secrets, in which case a chemical will only be listed as "…and other ingredients," Sutton adds. What are Chemical Equations? Detailed Explanation, Examples - BYJUS Chemical Equation: CaCl 2 + 2AgNO 3 → Ca (NO 3) 2 + 2AgCl↓ Ionic Equation: Ca 2+ + 2Cl - + 2Ag + + 2NO 3- → Ca 2+ + 2NO 3- + 2AgCl↓ Comparing the reactants and the products of the ionic equation and the chemical equation, it can be observed that the Ca 2+ ( calcium ion) and the NO 3- (nitrate) ions are present on both sides of the ionic equation.
Labeling A Chemical Equation Part 2 - YouTube The instructions for labeling a chemical equation.
7.3: Chemical Equations - Chemistry LibreTexts Use the common symbols, (s), (l), (g), (aq), and → appropriately when writing a chemical reaction. In a chemical change, new substances are formed. In order for this to occur, the chemical bonds of the substances break, and the atoms that compose them separate and rearrange themselves into new substances with new chemical bonds.
4.E: Chemical Reactions and Equations (Exercises) One element replaces another element in a compound. Zn + Fe (NO 3) 2 → Zn (NO 3) 2 + Fe 3F 2 + 2FeI 3 → 3I 2 + 2FeF 3 Sn + H 2 SO 4 → SnSO 4 + H 2 2Al + 3NiBr 2 → 2AlBr 3 + 3Ni No reaction occurs. Fe (NO 3) 3 + Al → Al (NO 3) 3 + Fe 2NaI + Cl 2 → 2NaCl + I 2 No reaction occurs. Zn (NO 3) 2 + 2NaOH → Zn (OH) 2 + 2NaNO 3 2HCl + Na 2 S → 2NaCl + H 2 S
How do you label a chemical equations? - Answers Chemical equations represent chemical composition of reactants and products and also how does the reaction occur. Given chemical equations be able to balance them? When one is given the chemical ...
Label the components of a chemical_ equation: - YouTube Label the components of a chemical_ equation:Watch the full video at: ...
























Post a Comment for "43 labels of a chemical equation"